|
12/9/2023 0 Comments Ap chemistry reference table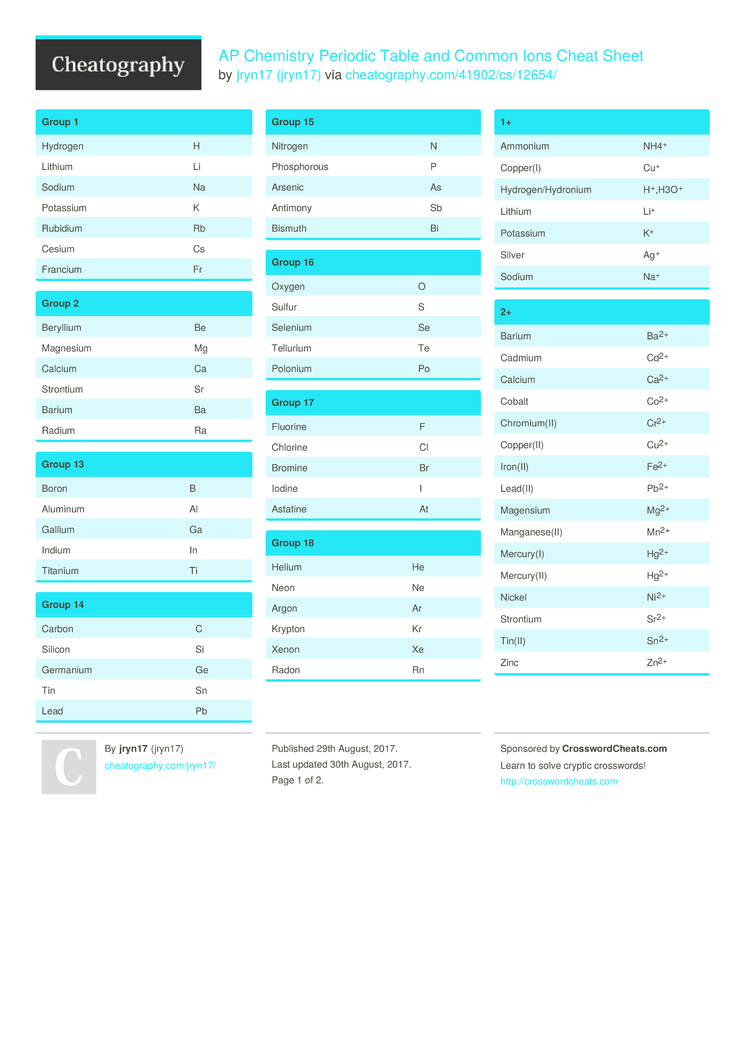
Very Good WebElements Interactive Periodic Table. If having more protons in the nucleus makes atoms smaller, why then, do they become larger as one moves Down a Column? As one moves down the column from one row to another, the distance to the outer electron shell increases.Ī Brief History of the Development of the first Periodic Table Additional Web Sites: Interactive Periodic Table. The outer electron feels a stronger pull. Why, then, do they become smaller?! Nuclear Charge increases while the outer electron shell (distance) remains the same. As distance increases force decreases a lot!! As one moves across a period, the atoms have more protons, more neutrons and more electrons. Force is proportional to 1/d2 (inverse squared). Why do atoms have a weaker hold on outer electrons as one moves Down a Column? Although the nuclear charge increases, the outer electrons are one shell farther from the nucleus. Why do the atoms have a stronger hold on outer electrons as one moves Across a Period (row)? The number of Protons or Nuclear Charge increases while the outer electrons are in the same shell or energy level (Bohr model). How do you know? Highest 1st Ionization Energy = Strongest hold and visa versa Indicate the location of the atoms with the strongest hold and the atoms with the weakest hold on their outer electrons. 
Table S and the two previous graphs and Illustrate the same trends going Down the Columns at the two ends of the Table. Strongest Hold on Outer Electrons 1st Ionization Energy Increases 1st Ionization Energy Decreases 1st Ionization Energy Decreases Atomic Radius Decreases Atomic Radius Increases Atomic Radius Increases Weakest Hold on Outer Electrons Consult Ref. Illustrate the trends in 1st Ionization Energy and Atomic Radius on the blank Periodic Table below. increases, the atoms become smaller!? Periodically, this trend also “starts over again.” How does the Periodic Table show this periodic character? Once again, the atoms that start and end the trend also start and end the Rows! State the general trend and describe the periodic character. How does the Periodic Table show this periodic character? The atoms that start and end the trend also start and end the Rows called Periods. increases 1st Ionization Energy also increases, but periodically this trend “starts over again”. 
It tells how strongly an atom attracts its outermost electrons. What is 1st Ionization Energy and what does it tell us about an atom? 1st Ionization Energy is the Energy required to remove one mole of electrons from one mole of atoms (Thus, each atom becomes an ion). How does the Periodic Table of Elements illustrate Periodic Trends in Physical and Chemical Properties? Use Reference Table S to examine periodic trends in 1st Ionization Energy and Atomic Radius when compared with atomic number. What are some examples of natural, periodic trends? Moon phases, ocean tides, water temperature in the northeast…. What does Periodic mean? Recurring or reappearing at regular intervals. We are not going to be wasting time in class copying notes.AP Chemistry Review of The Periodic Table What is a Trend? A general direction in which something tends to move. It is YOUR responsibility to download and print them or copy them off the Internet before the class when they are needed. If you do the work, you cannot possibly fail this class, regardless of your scores on exams. Grading for the class is most heavily based on completion of lab work and written lab reports. There will be very little assigned homework, but you should be reading the textbook and trying the practice questions daily. If just showing up for class is enough work for you, then do not expect to get higher than a 1 on the exam. Like any college class, you get out of it what you put into it. 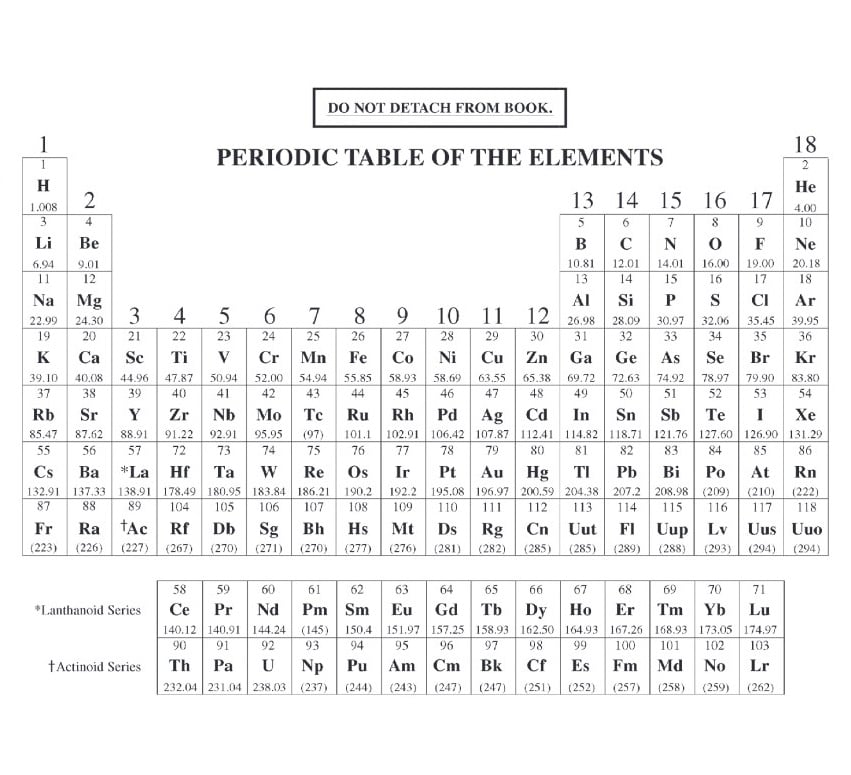
AP Chem has the reputation of being one of the hardest AP classes any student can take. Any student considering a career in Chemistry, Pharmacy, Medicine, Chemical Engineering, Biochemistry, or other related field is encouraged to take AP Chemistry. Those not getting college chemistry credit should find the first year chemistry class much easier than any student that hasn't taken AP Chem. The AP exam is given in early May and students earning a 3 or higher on the exam may get general chemistry, or another chemistry elective, waived. AP Chemistry is designed as a college freshman introductory or "General Chemistry" class.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
